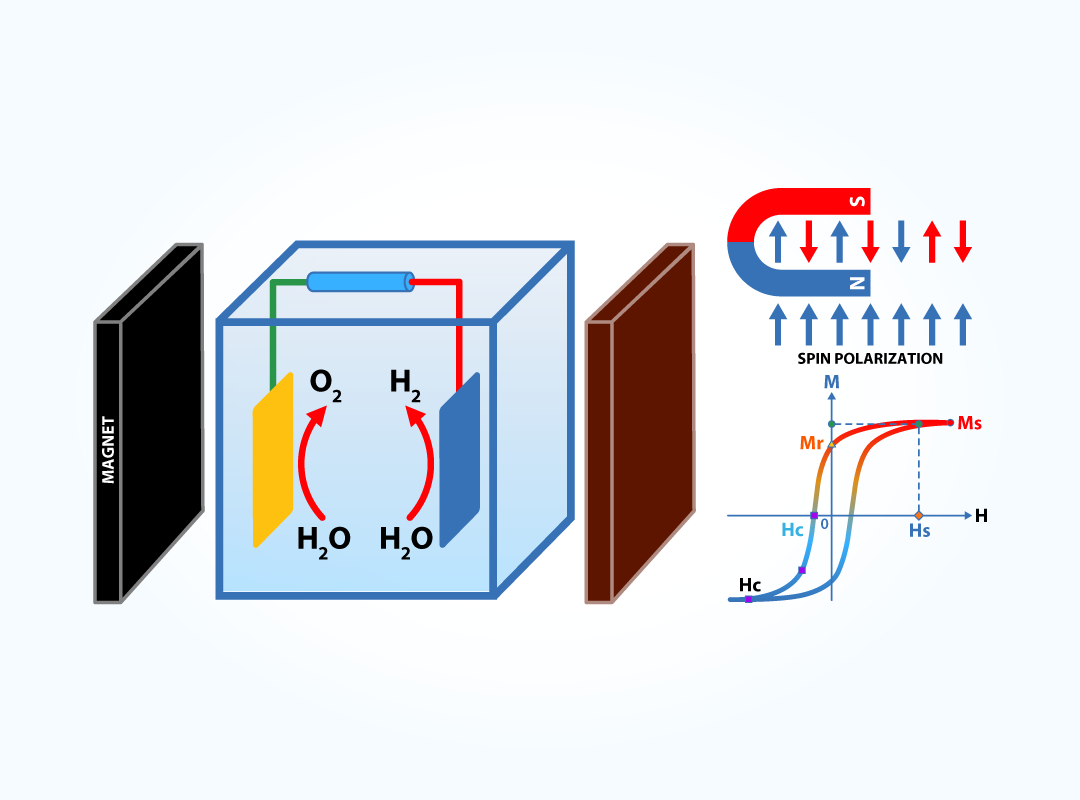
Magnetism drives energy conversion
Hydrogen production through alkaline water electrolysis, a key route to sustainable fuels, involves electron spin transitions, as a paramagnetic oxygen molecule is generated from diamagnetic species (H2O, OH⁻). This raises the intriguing question of how the magnetic ordering and spin state of catalysts influence oxygen evolution reaction (OER) kinetics. In our research lab, we study spin-dependent OER under external magnetic fields, and we aim to reveal its underlying mechanisms. Particularly, we focus on correlating the magnetic behavior of catalysts with OER performance, using spin- and magnetically-modified oxide and (oxy)nitride compounds. We also investigate the effect of catalyst spin states on chemisorption processes during the reaction. To gain deeper insights into the mechanisms, we employ in situ and operando techniques, including soft and hard X-ray absorption, resonant inelastic X-ray scattering, and X-ray magnetic circular dichroism, complemented by spin-polarized density functional theory (DFT) calculations. A magneto-electrochemical cell is developed for proof-of-concept studies. This research may address spin-related reaction barriers across diverse electrochemical processes.
Relevant Publications
[1] M. Hojamberdiev, K- Kawashima, M. Kumar, A. Yamakata, K. Yubuta, A. Gurlo, M. Hasegawa, K. Domen and K. Teshima, Engaging the flux-grown La1−xSrxFe1−yTiyO3 crystals in visible-light-driven photocatalytic hydrogen generation, International Journal of Hydrogen Energy, 2017, 42, 27024–27033.
Read the article
[2] M. Hojamberdiev, Y. Xu, F. Wang, W. Liu and J. Wang, La-modification of multiferroic BiFeO3 by hydrothermal method at low temperature, Inorganic Materials, 2009, 45, 1182–1186.
Read the article
[3] M. Hojamberdiev, Y. Xu, F. Wang, J. Wang, W. Liu and M. Wang, Morphology-controlled hydrothermal synthesis of bismuth ferrite using various alkaline mineralizers, Ceramics – Silikáty, 2009, 53, 113–117.
Read the article